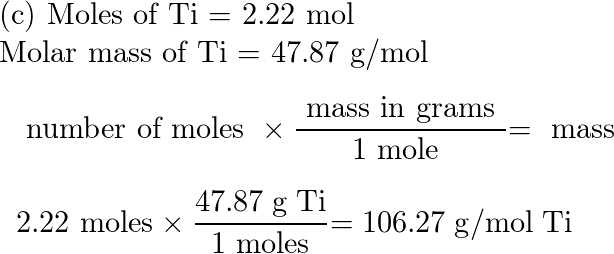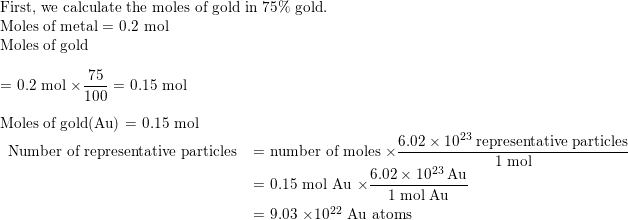Excited State Aromaticity and Antiaromaticity: Opportunities for Photophysical and Photochemical Rationalizations | Chemical Reviews

SOLVED:Jewelry A bracelet containing 0.200 mol metal atoms is 75% gold. How many particles of gold atoms are in the bracelet?
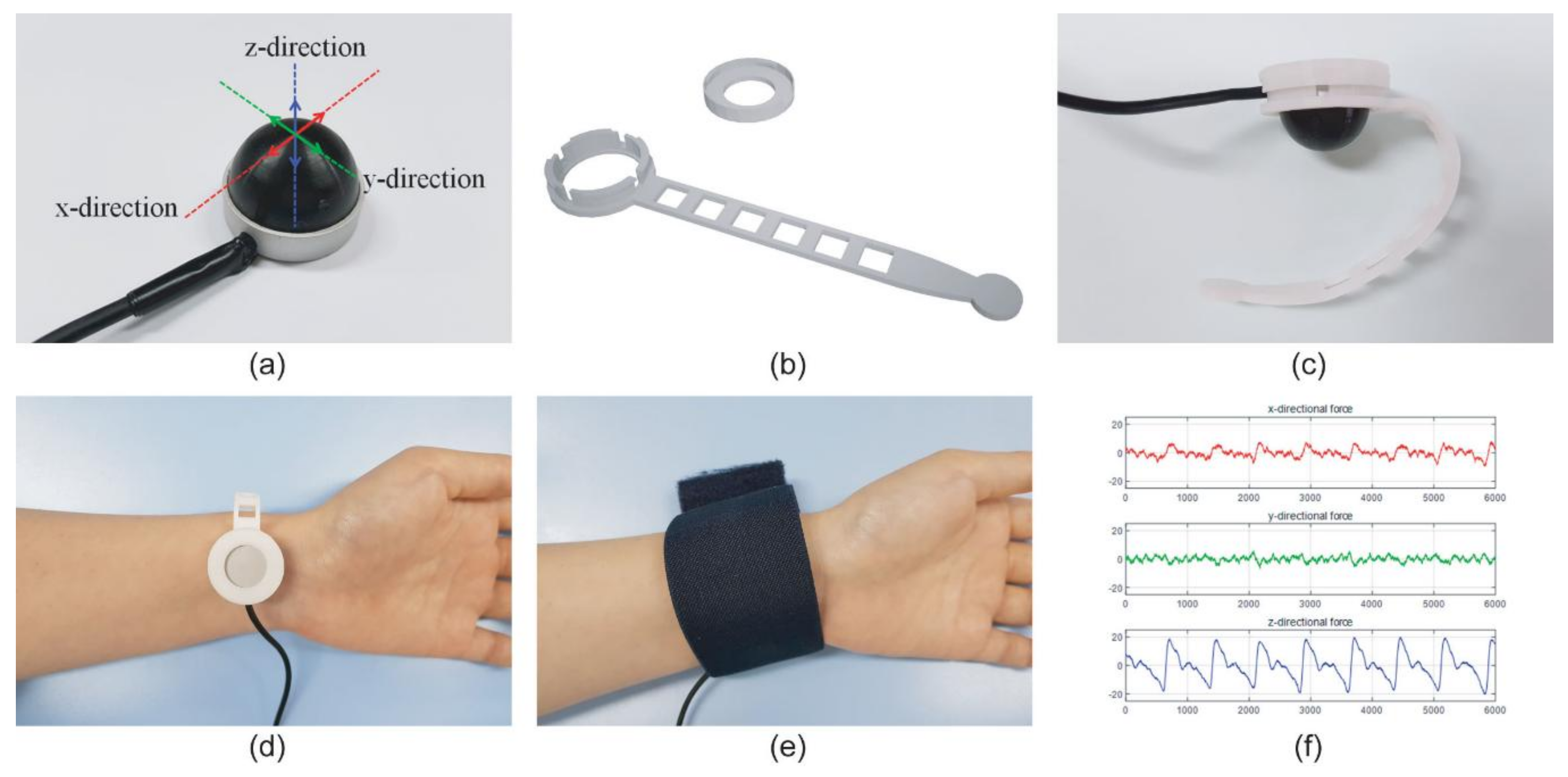
Sensors | Free Full-Text | Reliability and Validity of Non-invasive Blood Pressure Measurement System Using Three-Axis Tactile Force Sensor

SOLVED:Jewelry A bracelet containing 0.200 mol metal atoms is 75% gold. How many particles of gold atoms are in the bracelet?

SOLVED:Jewelry A bracelet containing 0.200 mol metal atoms is 75% gold. How many particles of gold atoms are in the bracelet?

SOLVED: Jewelry A bracelet containing 0.200 mol metal atoms is 75% gold. How many particles of gold atoms are in the bracelet?

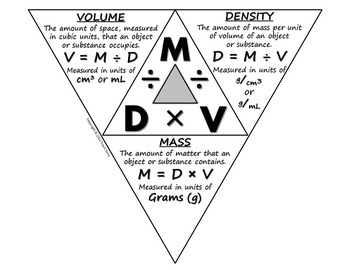
2 2 Calculations Involving the Mole.pdf - Calculations Involving the Mole 2.2 When making chocolate chip cookies it is more convenient to add a | Course Hero