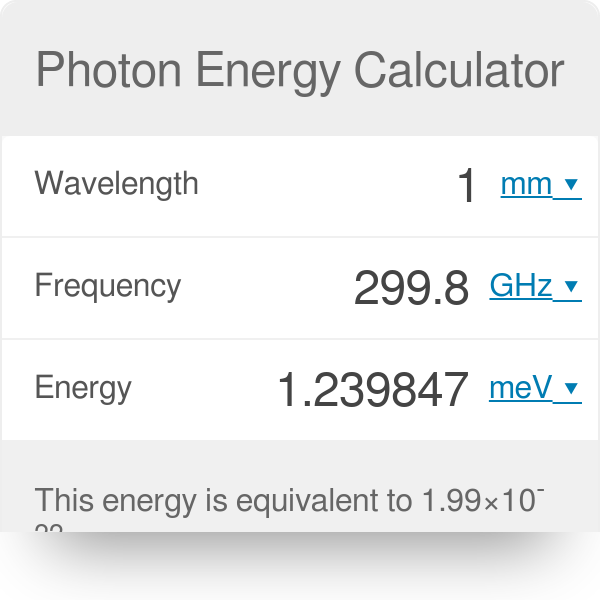
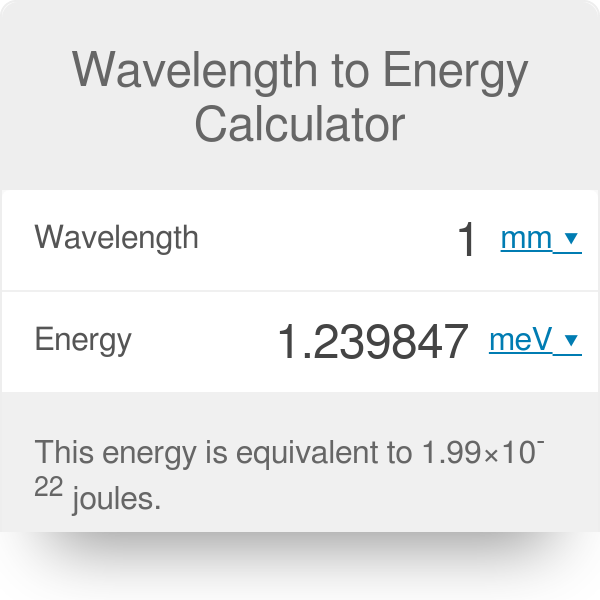
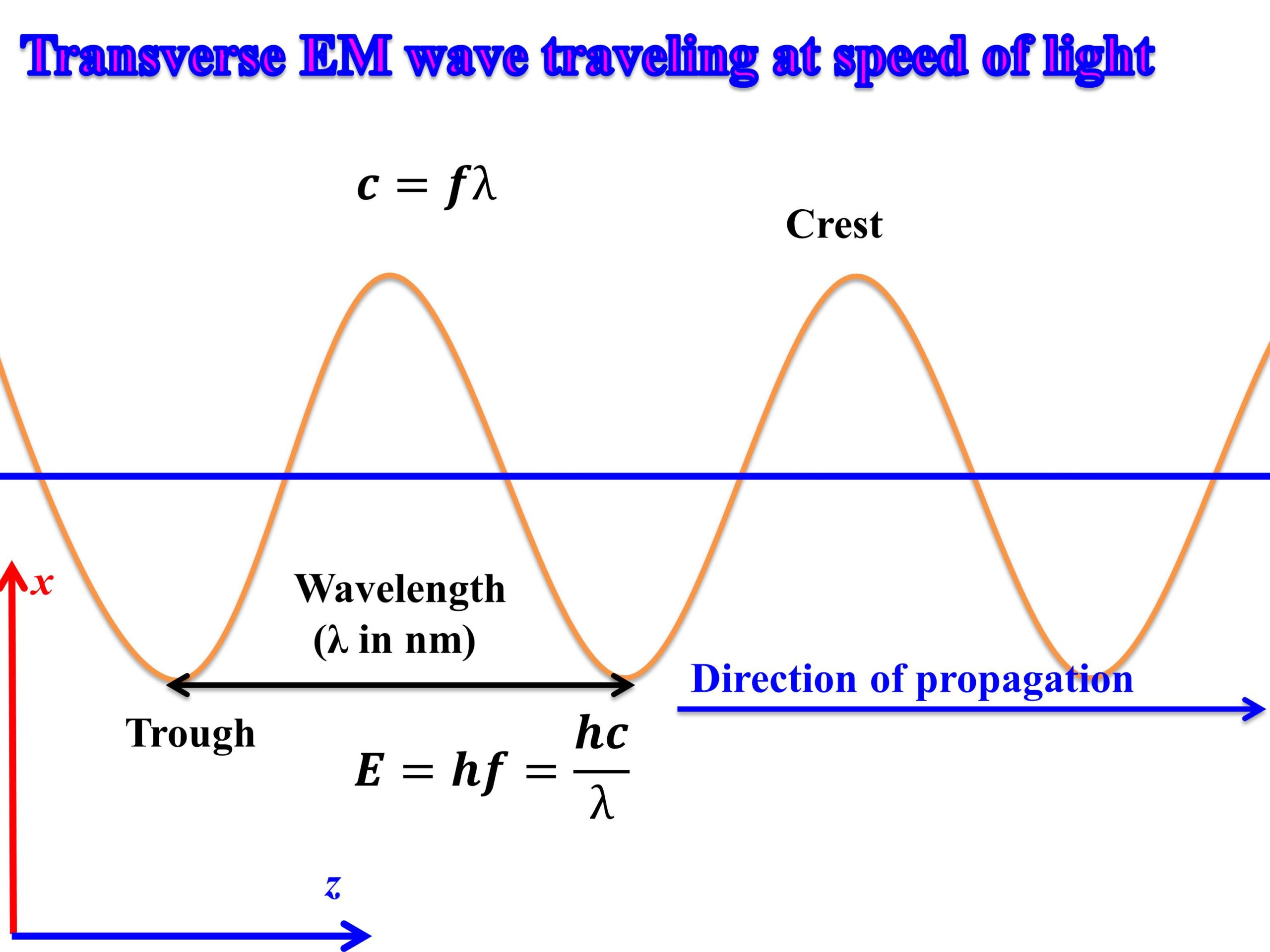
calculate the threshold frequency of the metal from which the photoelectrons are emitted with zero velocity when exposed to radiation of wavelength 6800 Armstrong

SOLVED: Constants Planck's constant: h = 6.626x10-34 J-s Speed of light: c = 3.00x108 m/s nm 10-9m Equations V=c/ Eph hv = hcl AE = E-Ei 22.179x10-18 J (1/n2) Calculate the energy

Find energy of each of the photons which (i) correspond to light of frequency 3 × 10^15Hz .(ii) have wavelength of 0.50 ∘A .