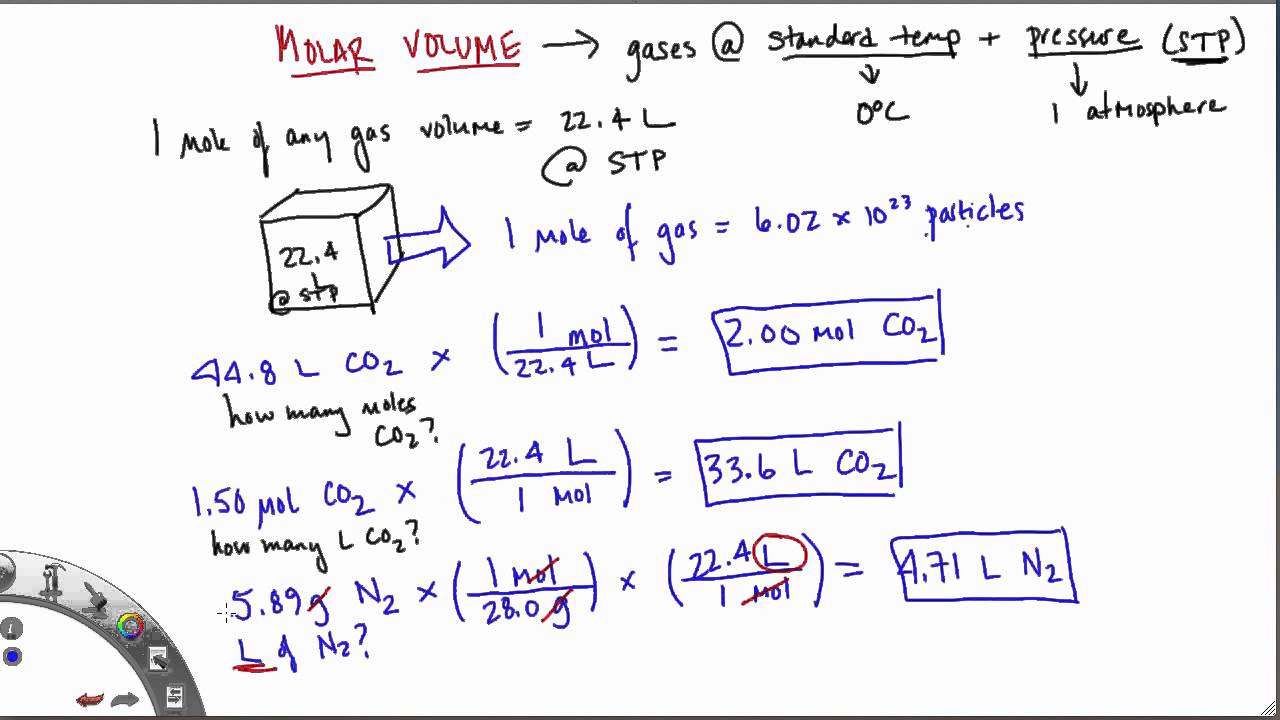
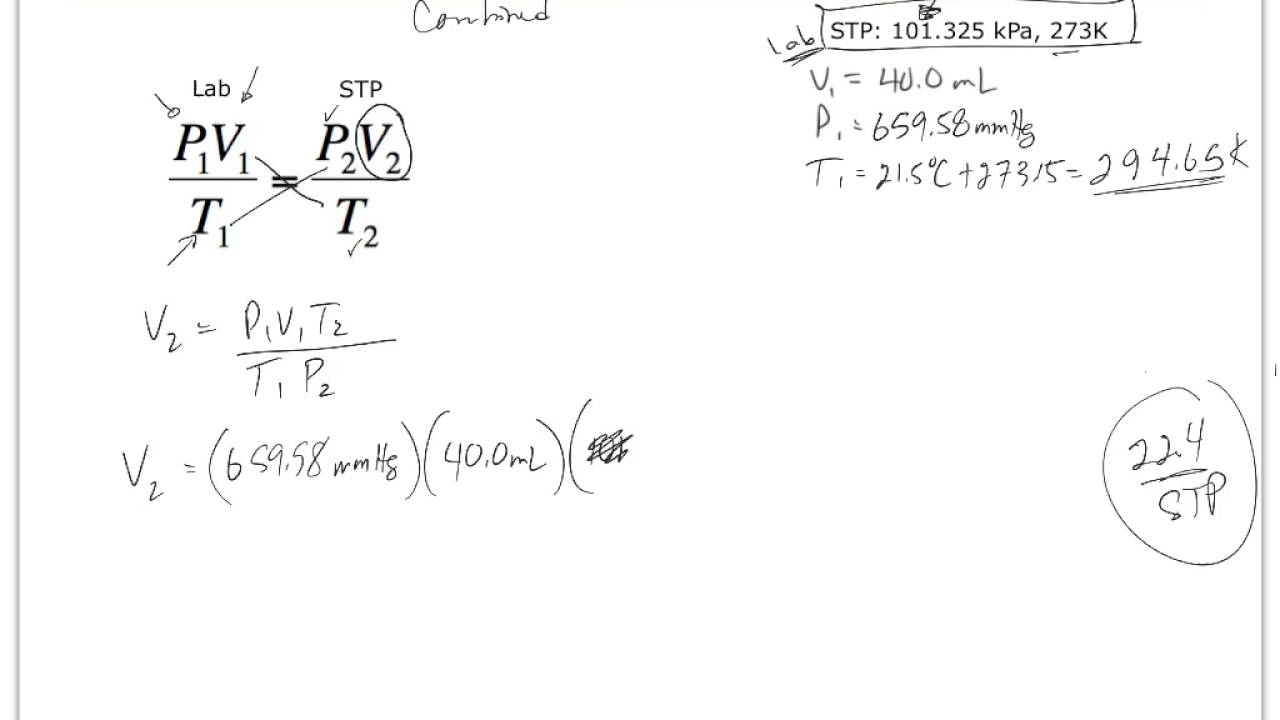Molar Volume Avogadro's Law: Equal volumes of gas at the same temperature and pressure contain equal numbers of particles. But the mass is different!!! - ppt download
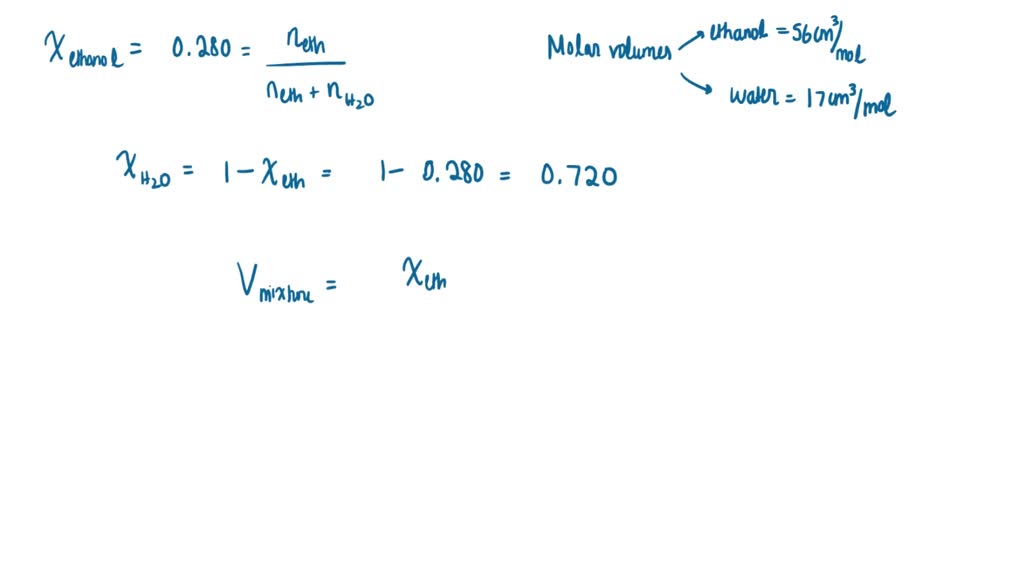
SOLVED: Calculate the volume of one mole (molar volume) of a mixture of water and ethanol in which mole fraction of ethanol is 0.280. Partial molar volumes of ethanol and water at

SOLVED:Use molar volume to calculate each of the following at STP: a. the volume, in liters, of 6.40 g of O2 gas b. the number of grams of H2 in 1620 mL

SOLVED:Calculate the molar volume of chlorine gas at 350 K and 2.30 atm using (a) the perfect gas law and (b) the van der Waals equation. Use the answer to (a) to

Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP : 1 atmospheric pressure, 0^oC ). Show that it is 22.4 litres .

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

Calculate standard molar volume of He ,if measurd density of He at stp is 0 1784g/l sir please solve - Chemistry - - 16212787 | Meritnation.com