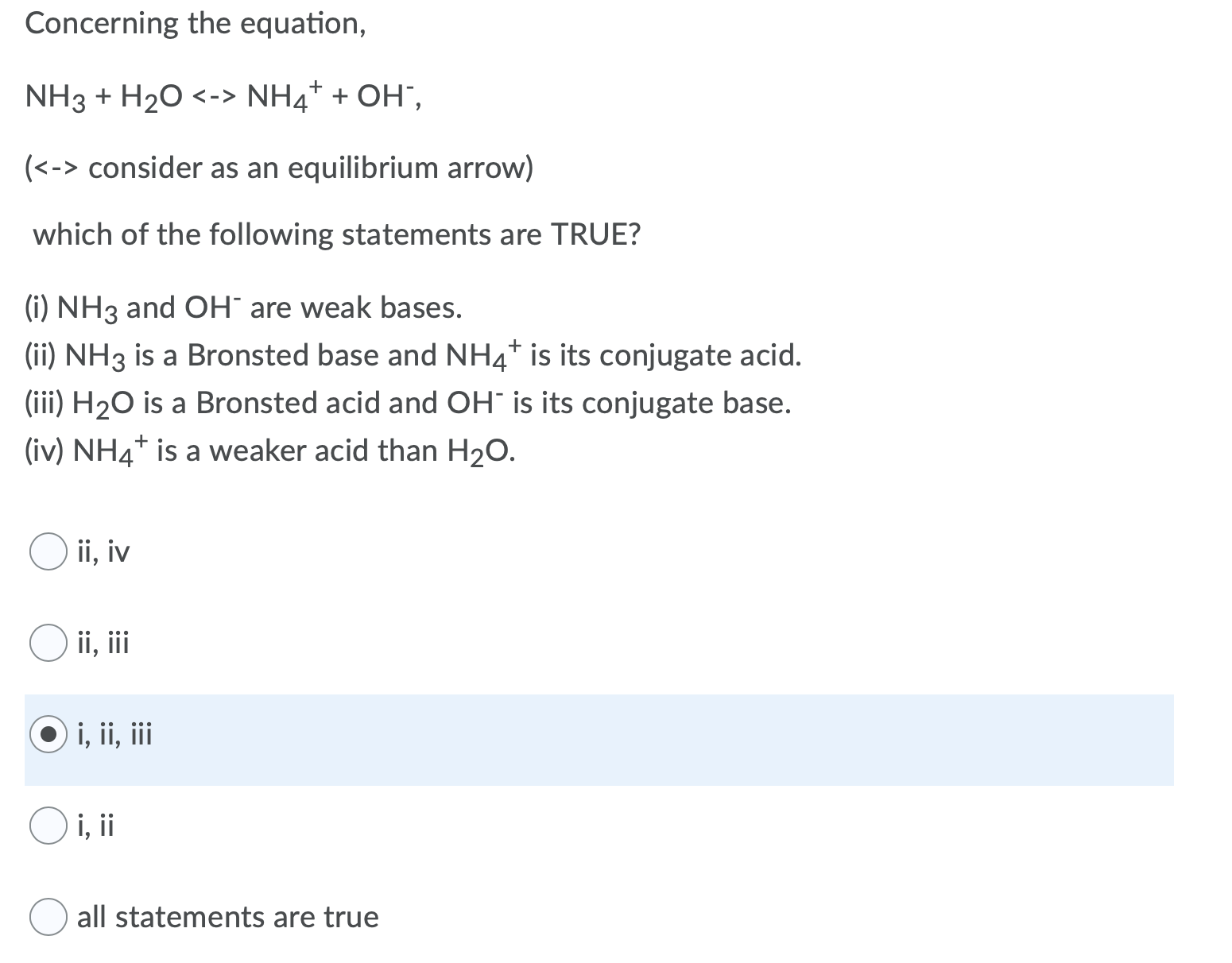
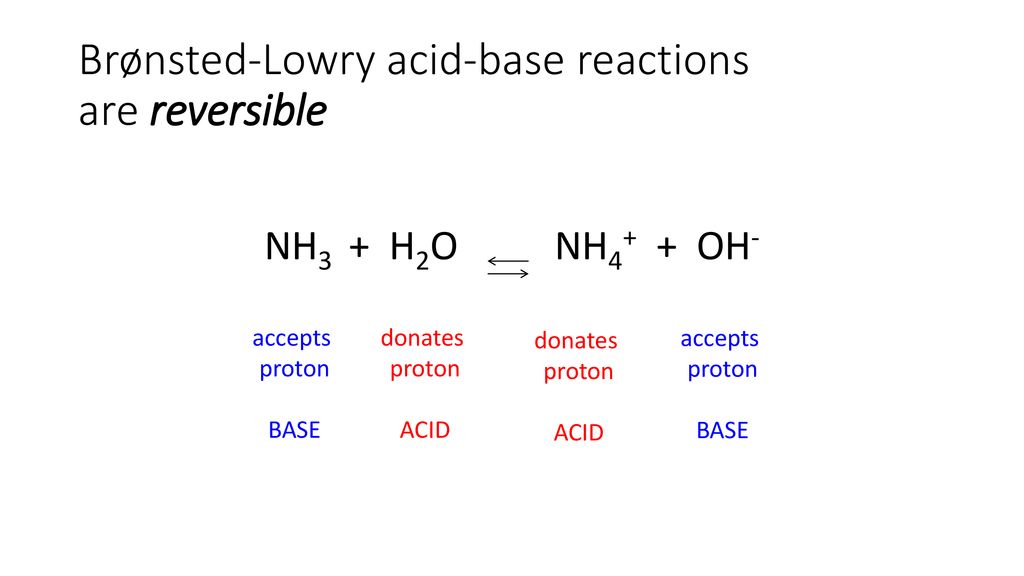
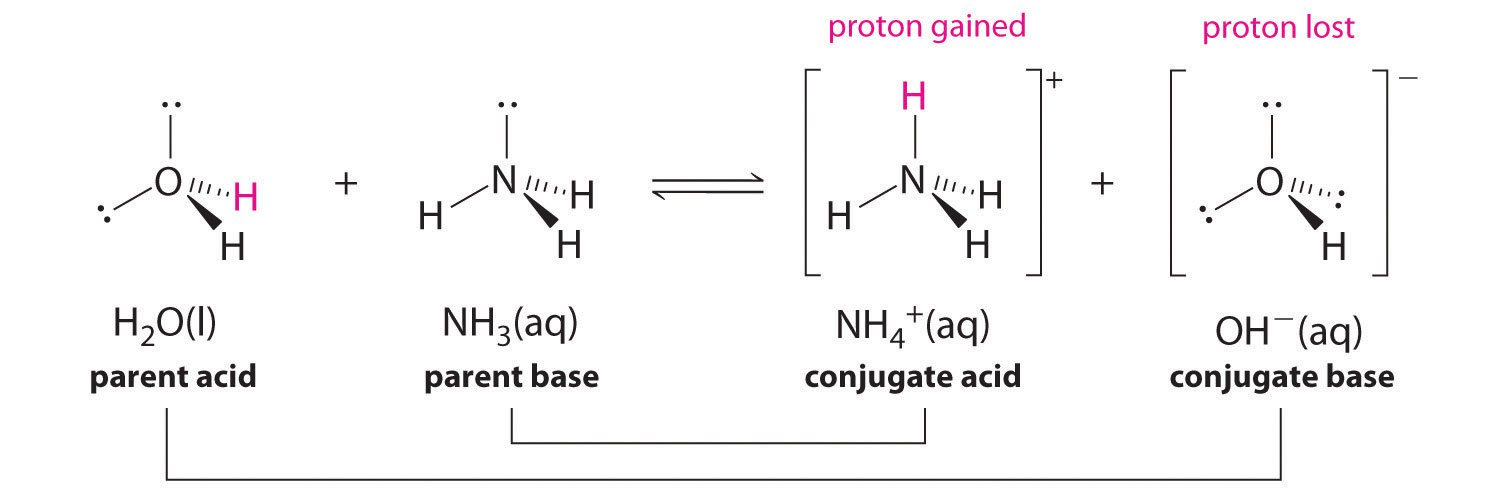
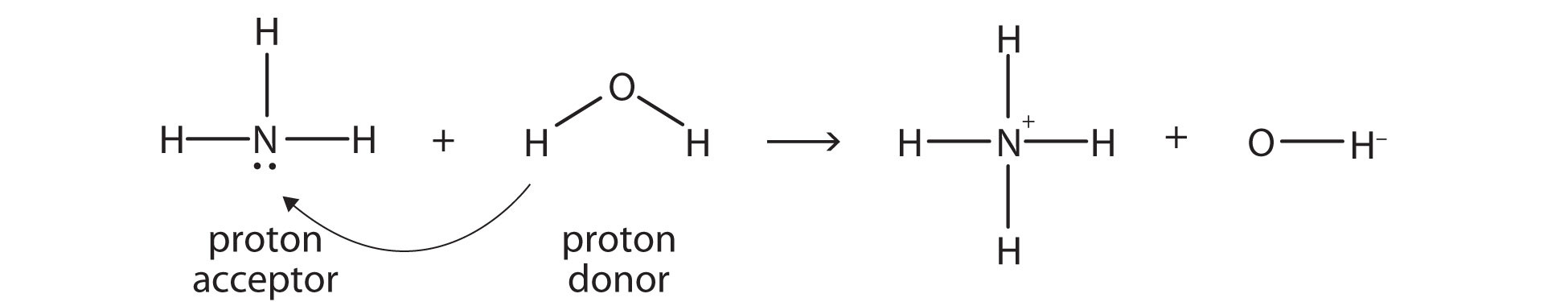
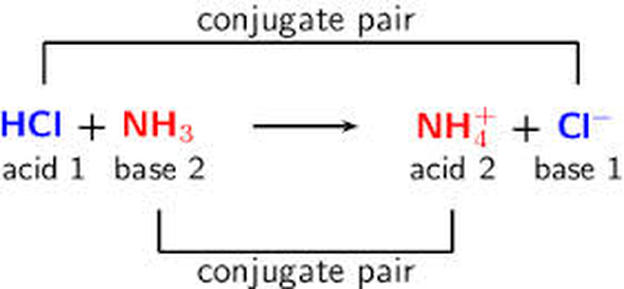
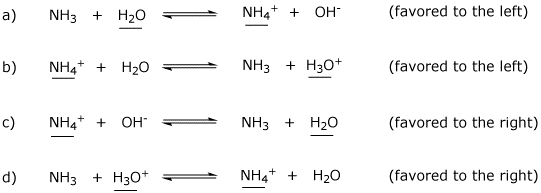
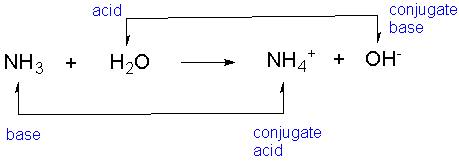
SOLVED: In the reaction NH3 (aq) + H2O (I) <–> NH4+ (aq) + OH- (aq), which is the conjugate acid-base pair? OH- NH3 NHA+, OH- NH3, H2O NH4+, NH3

Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com

SOLVED: Determine the conjugate acid/base pairs in the following reaction: NH4+ + OH- ⇔ NH3 + H2O A. NH4+/ H2O and NH3 /OH- B. NH4+/ NH3 and OH-/H2O C. Cannot be determined.

![The CO2-NH3-H2O system as described by the Thomsen model [7]. | Download Scientific Diagram The CO2-NH3-H2O system as described by the Thomsen model [7]. | Download Scientific Diagram](https://www.researchgate.net/publication/319196113/figure/fig1/AS:533643246669824@1504241872081/The-CO2-NH3-H2O-system-as-described-by-the-Thomsen-model-7.png)